Use Lomina fast test to detect neutralizing COVID-19 antibodies created in a body after an infection or after vaccionation.


IVD test designed for self-testing for the detection of IgG and IgM antibodies against S1 (RBD) protein in the blood after COVID-19 disease. The test is also designed to detect the approximate level of IgG antibodies after vaccination. The product does not serve as the primary tool for early detection of COVID19!





The test is developed and tested for the general public for the use of blood samples taken from a finger.

Whole blood
IgG – 96,79% (CI 95%: 94,46%-98,33%) / 97,27 %; (CI 95%: 95,86%-98,30%).
IgM – 98,54% (CI 95%: 97,14%-99,37%) / 88,46% (CI 95%: 84,18%-94,92%).
100% responsiveness to complex range of COVID-19 IgG antibodies
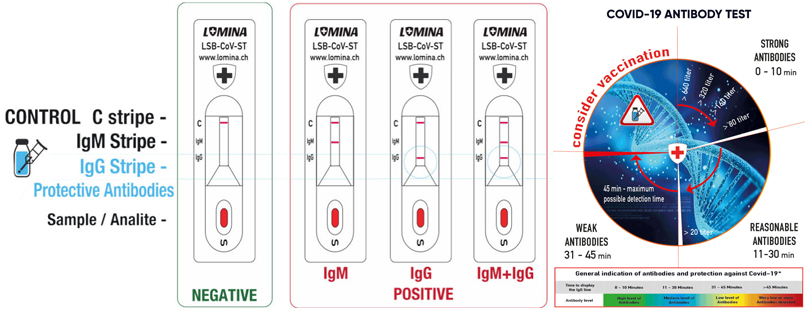
If the control strip QCC (Quality Control – C) is visible and lines M and G do not stain, the sample is negative because no antibodies were detected.
2.1. If control strip “C” is displayed together with the “IgM” strip, IgM antibody has been detected and the sample is positive. IgM antibodies indicate an acute reaction of the immune system after an immediate encounter with the virus, but it can also be triggered by a vaccine.
2.2. If the control strip “C” is displayed together with the “IgG” strip, an IgG antibody has been detected and the sample is positive. Depending on the display speed of the IgG band, it is possible to orientatively determine the value of antibodies after infection or after vaccination. The presence of IgG antibodies in the body is desirable to increase protection against COVID-19
2.3. If control strip “C” is displayed together with the “IgG” strip and the “IgM” strip, both IgG and IgM antibodies have been detected and the sample is positive for both types of antibodies.
If the check bar “C” does not appear, the test is invalid in any case and you must repeat it! This can be caused by a long test delay, reagent contamination, temperature degradation of the test, and the like.
As a globally active European brand, we distribute our portfolio of products to an extensive network of our business partners’ brick-and-mortar shops and e-shops.
| Cookie | Duration | Description |
|---|---|---|
| cookielawinfo-checkbox-analytics | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Analytics". |
| cookielawinfo-checkbox-functional | 11 months | The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". |
| cookielawinfo-checkbox-necessary | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". |
| cookielawinfo-checkbox-others | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. |
| cookielawinfo-checkbox-performance | 11 months | This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Performance". |
| viewed_cookie_policy | 11 months | The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. It does not store any personal data. |
